Cardiotoxicity Screening Market Size to Surpass USD 8.46 Billion by 2034
Cardiotoxicity Screening Market Size and Growth 2025 to 2034
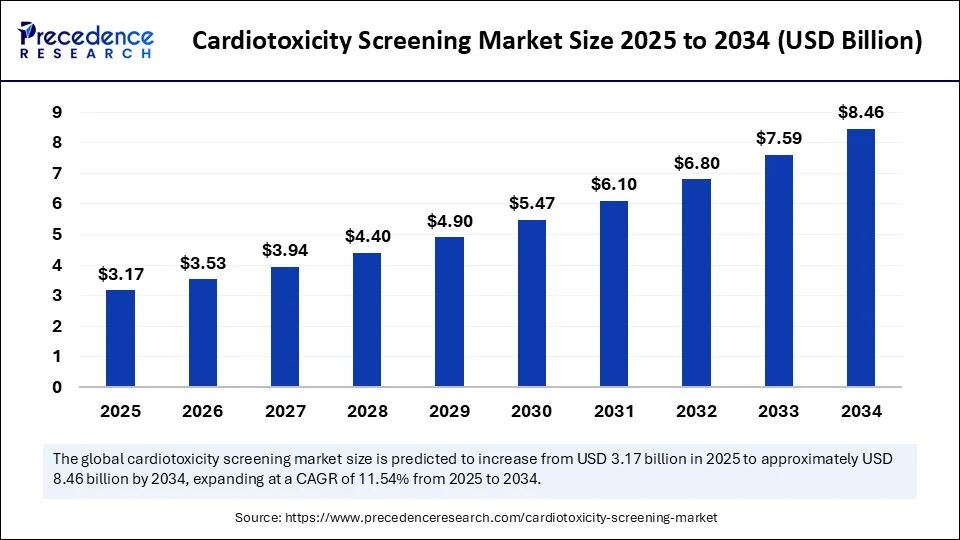
The global cardiotoxicity screening market is on a remarkable growth trajectory, poised to rise from USD 3.17 billion in 2025 to an impressive USD 8.46 billion by 2034. This growth reflects a robust compound annual growth rate (CAGR) of 11.54% fueled by increasing demand for safer, more precise cardiotoxicity screening technologies and expanding awareness of drug safety protocols.
The market’s key drivers include rising prevalence of cardiovascular diseases, technological advancements, especially in AI integration, and the growing influence of oncology therapeutics that require rigorous cardiac safety monitoring.
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @ https://www.precedenceresearch.com/sample/6624
What Are the Quick Insights Driving This Market’s Expansion?
-
The market size was USD 2.84 billion in 2024 and is expected to reach USD 8.46 billion by 2034.
-
North America holds the dominant share due to advanced healthcare systems and regulatory frameworks.
-
Asia Pacific is the fastest-growing region, propelled by increasing healthcare expenditure and innovation in screening technologies.
-
The U.S. market alone is projected to exceed USD 2.65 billion by 2034.
-
Leading industry players include Molecular Devices, Creative Bioarray, Miltenyi Biotech, and Axol Bioscience.
-
High-growth assay segments include hERG/Kv11.1 binding and patch-clamp assays, while the multi-electrode array with iPSC-cardiomyocytes is the fastest growing.
-
The pharmaceutical and biotechnology companies segment dominates end-user adoption, investing heavily in in-house screening technologies.
What Is Driving the Cardiotoxicity Screening Market’s Rapid Growth?
The market is growing due to several converging growth factors. Rising cardiovascular diseases worldwide have intensified the demand for early and accurate cardiac safety testing. Advances in cardiotoxicity screening technology, such as high-throughput screening, in vitro human-relevant models (e.g., iPSC-cardiomyocytes), and computational silico models, enable faster and more predictive assessments. Furthermore, the increasing use of chemotherapy and oncology therapeutics, which carry significant cardiac risks, propels the adoption of cardiotoxicity screening to enhance patient safety.
How Is Artificial Intelligence Transforming Cardiotoxicity Screening?
Artificial Intelligence (AI) is revolutionizing cardiotoxicity screening by improving the accuracy and efficiency of early detection. AI algorithms analyze complex ECG data and predict drug-induced cardiotoxicity, supporting safer drug development and clinical practices. This technology plays a pivotal role in cardio-oncology, where AI helps mitigate the risk of treatment-induced heart damage.
Additionally, AI-enabled tools facilitate pro-arrhythmia predictions and enable the integration of vast datasets for refined cardiac safety evaluations. This AI-driven transformation accelerates research efforts and supports regulatory compliance by providing more reliable and scalable cardiotoxicity assessments.
Cardiotoxicity Screening Market Scope
| Report Coverage | Details |
| Market Size by 2034 | USD 8.46 Billion |
| Market Size in 2025 | USD 3.17 Billion |
| Market Size in 2024 | USD 2.84 Billion |
| Market Growth Rate from 2025 to 2034 | CAGR of 11.54% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
| Base Year | 2024 |
| Forecast Period | 2025 to 2034 |
| Segments Covered | Assay/Test Type, Modality/Approach, Product and Service Type, End User, and Region |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Which Market Segments Lead and Why?
-
Assay/Test Types: The hERG (Kv11.1) binding and patch-clamp assays segment led in 2024, crucial for identifying fatal heart rhythm risks during drug development. Multi-electrode arrays with iPSC-cardiomyocytes present the fastest growth due to non-invasive, human-relevant screening benefits.
-
Modality: In vitro (cell and tissue-based) screening dominates, offering more accurate human model data compared to animal testing, especially leveraging hiPSC-CMs and 3D cultures.
-
Product & Services: Assay kits, reagents, and ready-to-use cells are essential for efficient, reliable drug safety testing. Contract research organizations (CROs) providing specialized screening services are the fastest-growing segment.
-
End Users: Pharmaceutical and biotechnology companies lead in-house screening activities, investing heavily in advanced technologies. CROs are increasingly outsourced for cost-effective, specialized cardiotoxicity screening.
Which Regions Are Pivotal in Market Growth?
-
North America: The leading region driven by advanced healthcare infrastructure, strict regulatory safety guidelines, and the presence of major pharmaceutical players. The U.S. fosters innovation through supportive policies like the FDA Modernization Act that promotes AI use and non-animal testing models.
-
Asia Pacific: The fastest-expanding region, bolstered by growing healthcare investments, technological innovation in 3D model cells, microfluidics, and AI analytics. China and India are key contributors, with China pioneering technological advancements driven by stringent regulations and growing R&D investments.
What Are the Latest Breakthroughs and Key Companies Shaping the Market?
Recent breakthroughs have centered on integrating AI, high-throughput screening, and human-relevant cellular models to improve precision and efficiency. Innovations in in vitro techniques like multi-electrode arrays with iPSC-cardiomyocytes and silico computational models have been front and center.
Key market players leading these advancements include:
-
Molecular Devices
-
Creative Bioarray
-
Miltenyi Biotech
-
Axol Bioscience
-
Cyprotex
-
Eurofins Discovery
-
Stemina Biomarker Discovery
-
Charles River Laboratories
-
IQVIA
-
Indigital Technologies
-
Labcorp Drug Development
What Market Challenges and Cost Pressures Exist?
Despite rapid growth, the market faces obstacles such as high capital expenditure for advanced instrumentation, variability in human cell protocols, and the translational gap between animal models and human physiology. Regulatory harmonization and the need for validated predictive algorithms remain critical barriers. Additionally, the substantial cost of cardiotoxicity screening technologies limits accessibility for smaller pharmaceutical companies and regions with constrained healthcare budgets.
Can Case Studies Showcase Successful Cardiotoxicity Screening?
In oncology, a leading pharmaceutical company successfully integrated AI-powered cardiotoxicity screening into chemotherapy drug trials. This early detection system significantly reduced cardiotoxic side effects, lowering patient hospitalization rates and improving therapeutic outcomes. This case underscores how combining advanced screening technologies with AI can optimize drug safety and efficacy.
Read Also: Spot Check Monitors Market
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344
